| Case Name |
Explosion caused due to heat of adsorption of an adsorption tower used to deodorize a methyl acrylate tank |
| Pictograph |
|
| Date |
March 9, 1976 |
| Place |
Himeji, Hyogo, Japan |
| Location |
Chemical plant |
| Overview |
A storage tank suddenly exploded during transferring from a rundown tank to a storage tank of methyl acrylate. Due to heat of adsorption and oxidation of active carbon in the deodorization tower, which prevented an odor from diffusing into the atmosphere, the vapor was ignited and there was a tank fire. No prior examination was made of the adsorption system of the adsorption tower. An inert gas blanket to prevent against the formation of explosion gas mixture was not applied to the tank either. |
| Incident |
During receiving methyl acrylate into a storage tank, a combustible gas-air methyl acrylate mixture existed in the tank was ignited and exploded by heat of adsorption and oxidation from activated carbon used as an absorbent in the adsorption tower for deodorization. The capacity of the tank was 1,000 kL. |
| Processing |
Storage |
| Substance |
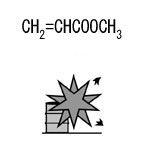
Methyl acrylate, Fig2 |
| Type of Accident |
Explosion, health hazard |
| Sequence |
The tank had been used for one week. By the day of the accident, a total of about 325 kL of methyl acrylate had been received four times, and at the time of the accident, about 100 kL remained in the tank.
10:40 on March 9th, 1976; it was confirmed that there were no problems at valves and gages of a product tank and storage tanks.
About 11:00; receiving started. Prior to this the temperature of a liquid in the tank was 15°C.
12:05; when about 17 kL of methyl acrylate were received, the storage tank exploded and a fire occurred. As a result of the explosion, an exterior iron plate of the tank, the ceiling, insulators, angles, indicators, etc were scattered over the range of 200 m. Part of the ceiling was blown off into the sea. After the accident, about 5 kL of liquid material were left in the tank. |
| Cause |
A combustible gas-air mixture of methyl acrylate was generated at the vapor layer of the tank and the deodorization tower. The ignition source was thought to be heat of adsorption and oxidation with activated carbon used as an absorbent in the deodorization tower. The deodorization tower has a diameter of 600 mm, and a height of 4 m. |
| Response |
Foam extinguishing was carried out by the company's fire brigade, and the public fire brigade, and water spraying was done for cooling. |
| Countermeasures |
The deodorization tower was changed from an adsorption system with active carbon to an alkali absorption system. In addition, an inert gas blanket to prevent against the formation of explosion gas mixture was provided at at the tank. |
| Knowledge Comment |
It is necessary to be careful about the generation of heat of adsorption when an adsorbent is used. An explosion gas mixture should not be generated in the tank. In the case, as methaquinone has no inhibition effect on polymerization in the absence of oxygen, an inert gas blanket containing ca. 7 vol.% of oxygen should be provided at the tank. |
| Background |
The cause was thought to be insufficient study on the adsorbent method at the methyl acrylate deodorization tower. An experiment at Himeji Institute of Technology after the accident clarified that when activated carbon adsorbed methyl acrylate, the temperature reached over 500°C because of the accumulation of adsorption and oxidation heat. |
| Incidental Discussion |
A nitrogen blanket was not adopted at the factories of other companies in the same business, because polymerization of acrylic ester occurs.
After the explosion accident an inert gas blanket containing ca. 7 vol.% of oxygen was introduced to prevent against the formation of the explosion gas mixture and the inert gas blanket method was adopted in "Safe handling guideline of acrylic acid and acrylic ester" published by Acrylic Acid Ester Industry Association. |
| Reason for Adding to DB |
Example of explosion of which the ignition source was clarified in a demonstration test |
| Scenario |
| Primary Scenario
|
Poor Value Perception, Poor Safety Awareness, Inadequate Risk Recognition, Insufficient Analysis or Research, Insufficient Prior Research, No Study about Heat of Adsorption,etc., Planning and Design, Poor Planning, Poor Design, Bad Event, Chemical Phenomenon, Heat generation, Secondary Damage, External Damage, Explosion/Fire, Bodily Harm, Injury, 57 person injured, Loss to Organization, Economic Loss, Manetary Damage 150 mollion yen
|
|
| Sources |
Fire and Disaster Management Agency. Examples of accidents at dangerous material manufacturers in 1976. pp. 128-129.
Himeji City Fire fighting station. Outline of explosion and fire at H factory N chemical industry.
Tetsuzo Kitagawa. Explosion at methyl acrylate tank. Analysis of the explosion hazard. pp.73-74,230(1980)
Fire and Disaster Management Agency. Fire at vent pipe with deodorization column caused by heat formation and ignition inside deodorization column. Examples of accidents at outdoor tanks. No.2. pp.17-29(1983).
|
| Number of Injuries |
57 |
| Physical Damage |
Inside the factory: Facilities attached to the tank burned and were damaged. Windows of buildings were broken by a blast.
Outside the factory: Windows of buildings within a 1.7 km radius were damaged (12 surrounding buildings, three company houses, one hospital, 19 houses). |
| Financial Cost |
¥ 150 million (Fire and Disaster Management Agency) |
| Consequences |
There was an irritating odor in the surrounding residential area, and residents complained. On March 10th, 1976, 60 employees investigated residents' health of 3000 families around the plant. |
| Multimedia Files |
Fig2.Chemical formula
|
| Field |
Chemicals and Plants
|
| Author |
WADA, Yuji (National Institute of Advanced Industrial Science and Technology)
TAMURA, Masamitsu (Center for Risk Management and Safety Sciences, Yokohama National University)
|
|